Aluminium toxicity
-
Aluminium and soil acidity
-
Effects on plants and mechanisms of adaptation
-
Recommended reading: Brady and Weil, 2002 and Marschner,
1995.
1. Aluminium and soil acidity
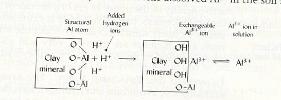 |
| Fig.1 Adsorbed and exchangeable Al-ions |
| (
Source: Brady and Weil, 2002.) |
-
Aluminum (Al) is a major constituent of most soil minerals
(Al-silicates, Al-Oxides). When the soil becomes more acid, more
H+ ions are present which adsorb on a clay surface.
They usually do not remain as exchangeable cations for long but react with the
primary minerals. Al is released from the lattice structure and becomes
adsorbed at the surface of the clay minerals.
-
These exchangeable Al-ions, in turn, are in equilibrium with
the soil solution (see fig.1 above).
-
The exchangeable and dissolved
Al3+ ions (Al3+ =
Al(OH2)63+)
play two critical roles in the soil acidity stay:
-
Al is toxic for most organisms
-
Reacts with water (hydrolyzes) to form H-ions. A single
Al can release 3 H-ions (on this account, Al and H are referred to as acid
cations)
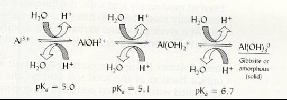 |
| Fig.2 Al reaction with water |
| (
Source: Brady and Weil, 2002.) |
-
A general relationship between the soil pH and the solubility
of Al compounds is shown in fig.3 below.
 |
| Fig.3 Aluminium as a function of pH |
| (
Source: Marschner, 1995.) |
-
Al-toxicity is not a problem when pH > 5.2
(pH-CaCl2 > 4.8) because little free Al or
Al-hydroxide (AlOH2+) is in the soil solution.
Al-toxicity to plants is often greatest near pH 4.5 because the
AlOH2+ species which is more toxic than the
Al3+ species is most soluble at that pH.
 |
| Fig.4 Al in the soil solution |
| (
Source: Brady and Weil, 2002.) |
-
Effects of Al3+ ions in the soil
solution of acid mineral soils below a pH of 5.5 are
-
Increasing proportion of the cation exchange sites of
clay minerals is occupied by Al where it especially replaces other polyvalent
cations (Mg2+,
Ca2+).
-
Acts as a strong adsorber of phosphate and
molybdate.
2. Effects on plants and mechanisms of
adaptation
Effects on plants:
-
stunted root system, short, thick, root tips turn brown
 |
| Fig.5 Al-toxic roots |
| (
Source: http://www2.ctahr.hawaii.edu/ctahr2001/Soil/) |
-
Blocks sites where Ca is taken up
-
Binds to P (ATP, DNA)
Mechanisms of adaptation of plants in acid mineral
plants:
-
Al-tolerance is the most important individual factor required
for adaptation of species and cultivars to acid mineral soils.
-
Al-tolerant plants:
-
Exclude Al from their roots by raising the pH of the soil
just outside the root. This causes the Al to precipitate.
-
Excrete organic mucilage that complexes with Al.
-
Excrete organic acids that combine with Al.
-
Includers: achieved by accumulation in plant tissue. In
tropical rainforest up to 4780 mg Al per liter were found in the leaf press
sap; in old leaves of tea up to 30 mg per gram dry weight.
-
Exclusion from sensitive sites.
-
pH increase in the rhizosphere.
-
Root exudates: mucilage and organic acids bind and complex
Al.
-
Mycorrhizas: release of organic acids.
-
A summary of possible mechanisms of adaptation is shown in
fig.6 below.
 |
| Fig.6 Strategies of plant adaptation to acid
mineral soils |
| (
Source: Marschner, 1995.) |
|

 previous | next
previous | next